ONE IN FIVE WOMEN WILL DEVELOP A PELVIC MASS
Offering Protein Biomarker and Genetic Testing options to provide greater physician insights, facilitate early detection and optimize treatment plans.
- Over 10 years of experience in Ovarian Cancer Risk assessment and Pelvic mass management
- Cutting edge bioinformatics for analysis and data management
- Experienced in quality-focused patient care
- Laboratory Research focused on innovative discovery
We're on a mission to end late-stage ovarian cancer diagnosis. See how Aspira Women's Healths' products compare to CA-125
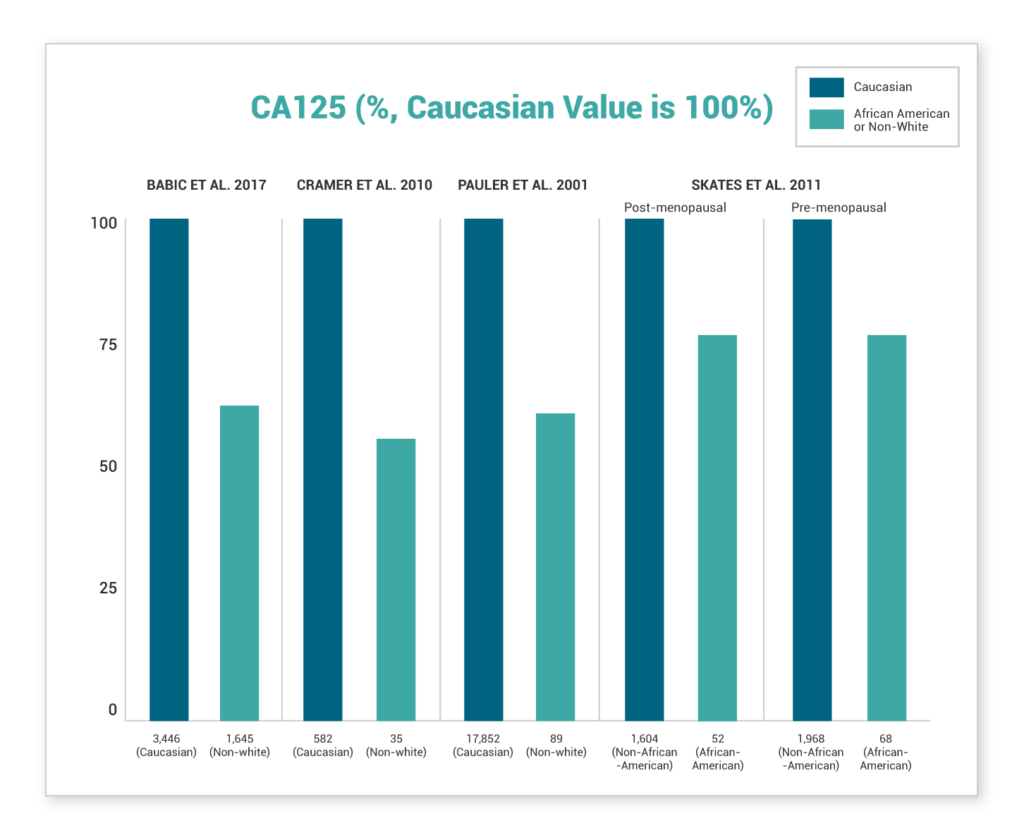
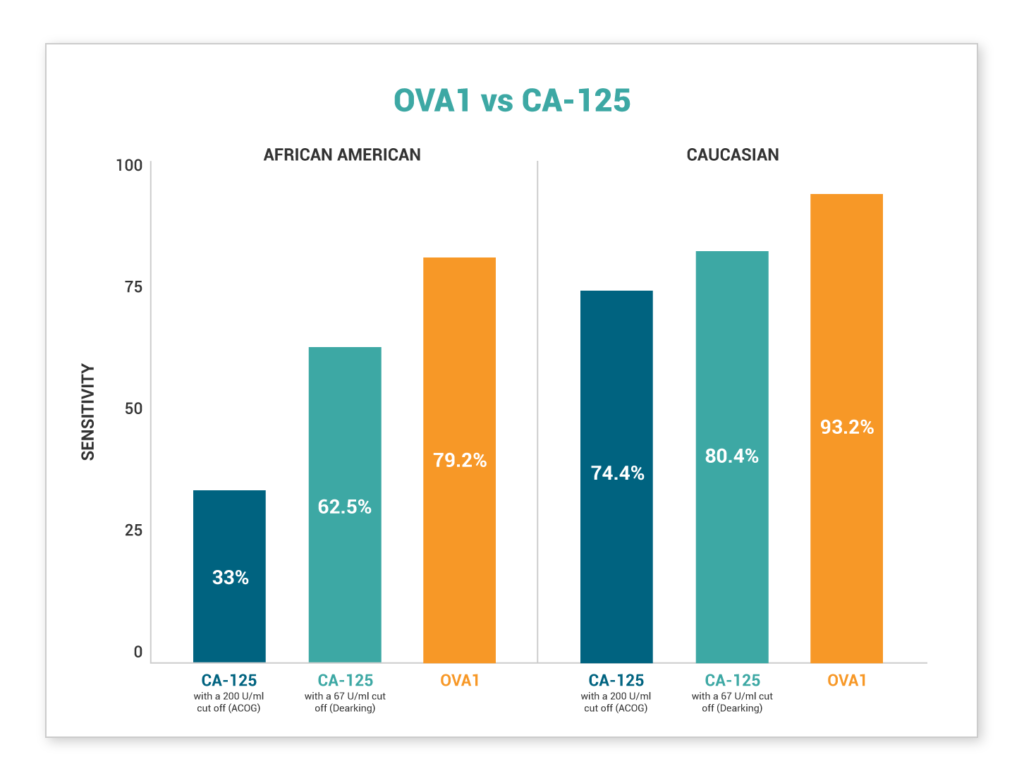
HELP US END LATE-STAGE OVARIAN CANCER DIAGNOSIS
Explore Our Products

OVA1plus
OVA1plus is a reflex process which performs OVA1 and OVERA, both FDA-cleared tests for women with adnexal masses. OVA1plus helps you further stratify your patient’s risk of malignancy for women with pelvic masses planned for surgery. Benefits of this enhanced test offering include; Reducing the falsely elevated rate by up to 50%, maintaining the high rate of detection (sensitivity) for ovarian cancer risk, continuing to provide confidence in low-risk results with a high negative predictive value, and increased specificity by up to 20%.

GenetiX
With a focus on women’s health, Aspira Labs® offers both comprehensive and targeted genetic testing options for Hereditary Breast and Ovarian cancer as well as Carrier Screening to help guide medical management for your patient.

Synergy Technology Transfer
Aspira Synergy allows labs to significantly reduce the time needed to implement in-house testing solutions
Developed by a team of experts, Aspira Synergy informs medical choices for women’s health with the assistance of multi-marker proteins and state-of-the-art genetic testing for both carrier screening and hereditary cancer risk.