
Part of the OvaSuiteSM portfolio of blood tests, Ova1Plus® is a combination of two FDA-cleared tests for women with pelvic masses who are planned for surgery.

Ova1® is performed first, leveraging its high sensitivity1. Overa®, with its high specificity2, is automatically performed when an intermediate risk is detected.
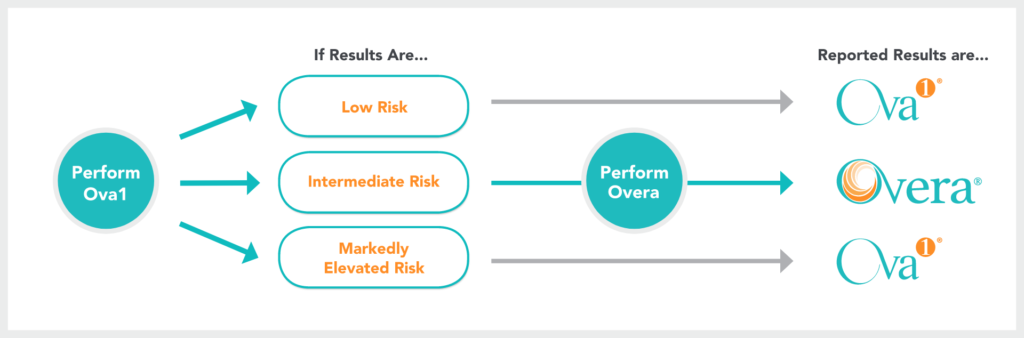
Low Risk is <4.4 (Postmenopausal) and <5.0 (Premenopausal) Intermediate Risk is 4.4-6.0 (Postmenopausal) and 5.0-7.0 (Premenopausal) Markedly Elevated Risk is >6.0 (Postmenopausal) and >7.0 (Premenopausal)

Learn more about Ova1’s high performance compared to CA 125 alone!
With 4 additional biomarkers and a proprietary algorithm, Ova1 has dramatically improved performance compared to CA 125 alone.

Ova1 demonstrates improved performance across certain ethnicities, compared to CA 125 alone.4

Biomarkers in Ova1 & Overa
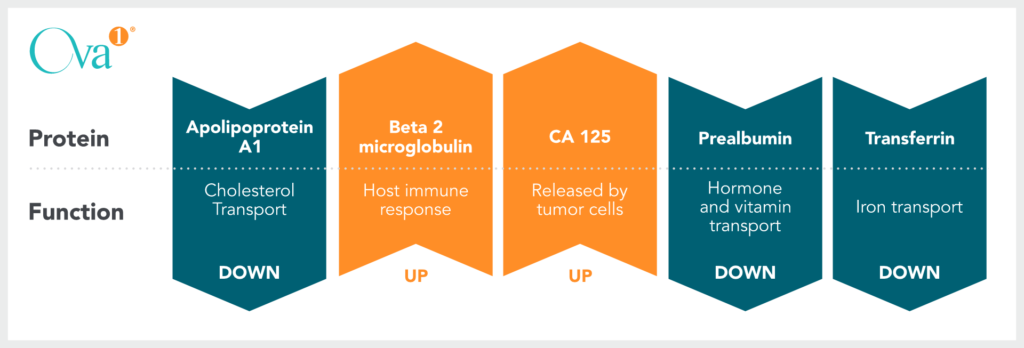
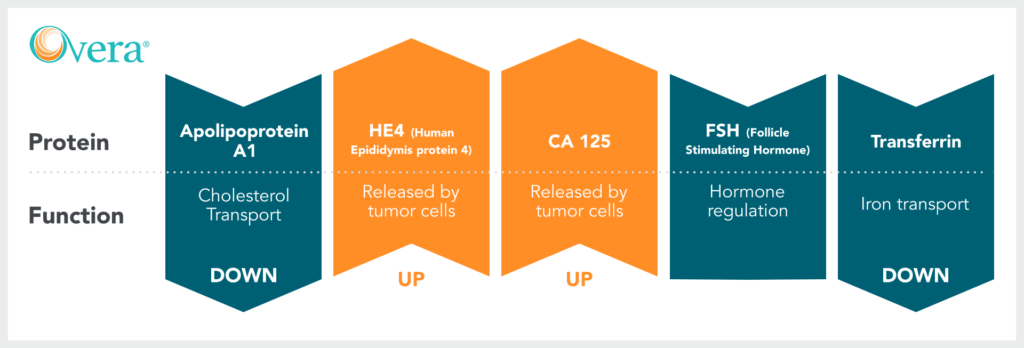
Arrows indicate the typical trends of biomarkers in the case of an Elevated Risk result.
Not planning for surgery?

Utilize the OvaSuite portfolio of products and confidently move from assessment to action.
A single blood draw – which can be performed at a number of national and regional laboratories or in a healthcare provider's offices using our proprietary testing kit – is all that is needed.
Contact us to learn more, order kits, or to locate one of our phlebotomy partners.
- Ovarian malignancy risk stratification of the adnexal mass using a multivariate index assay. Gynecologic oncology, 128(2), 252-259.
- Dunton, C. J., Bullock, R. G., Fritsche, H. A., & Twiggs, L. B. (2020). Improvement in MIA (OVA1) testing for detection of ovarian malignancy. Gynecologic Oncology, 159, 25.
- Dunton, C. J., Hutchcraft, M. L., Bullock, R. G., Northrop, L. E., & Ueland, F. R. (2021). Salvaging detection of early-stage ovarian malignancies when ca125 is not informative. Diagnostics, 11(8), 1440.
- Dunton, C., Bullock, R. G., & Fritsche, H. (2019). Ethnic disparity in clinical performance between multivariate index assay and CA125 in detection of ovarian malignancy. Future Oncology, 15(26), 3047-3051.
